Scientists at The Picower Institute for Learning and Memory at Massachusetts Institute of Technology (MIT) used an innovative microscopy method to observe how new-born neurons struggle to reach their proper locations in advanced human brain tissue models of Rett syndrome -bringing new understanding to how the brains of people with the potentially destructive illness have been found to have growing impairments.
Mutations in the gene MECP2 cause Rett syndrome, which is characterised by symptoms such as severe intellectual disability and impaired social behaviour.
Researchers grew 3D cell cultures called cerebral organoids, or mini-brains, using cells from people with MECP2 mutations and compared them to otherwise-identical cultures without the mutations to gain new insight into how the mutation affects the brain develops early on.
The development of each type of mini-brain was subsequently examined by the researchers, under the direction of postdoctoral fellow Murat Yildirim, using third harmonic generation (THG) three-photon microscopy. THG enables very high-resolution imaging deep into living, intact tissues without the use of chemicals to label cells.
The new study is the first to photograph organoids using THG, essentially leaving them undisturbed. Prior organoid imaging investigations necessitated the use of imaging technology incapable of penetrating the 3D tissue or techniques that necessitated killing the cultures by either slicing them into thin sections or chemically cleaning and labelling them.
Since a laser is used in three-photon microscopy, the lab’s microscope was specifically designed to only use as much power as a cat toy laser pointer on the tissue (less than 5 milliwatts).
The reason why researchers are so cautious regarding power and chemical labelling is so they can ensure they are not negatively altering or impacting the neuronal physiology and preserve everything intact without introducing anything external that may be harmful.
They managed to get enough signal, even at low power, to image fixed and living organoids without labels. They compared their THG images to those created using more conventional chemical labelling techniques to verify that.
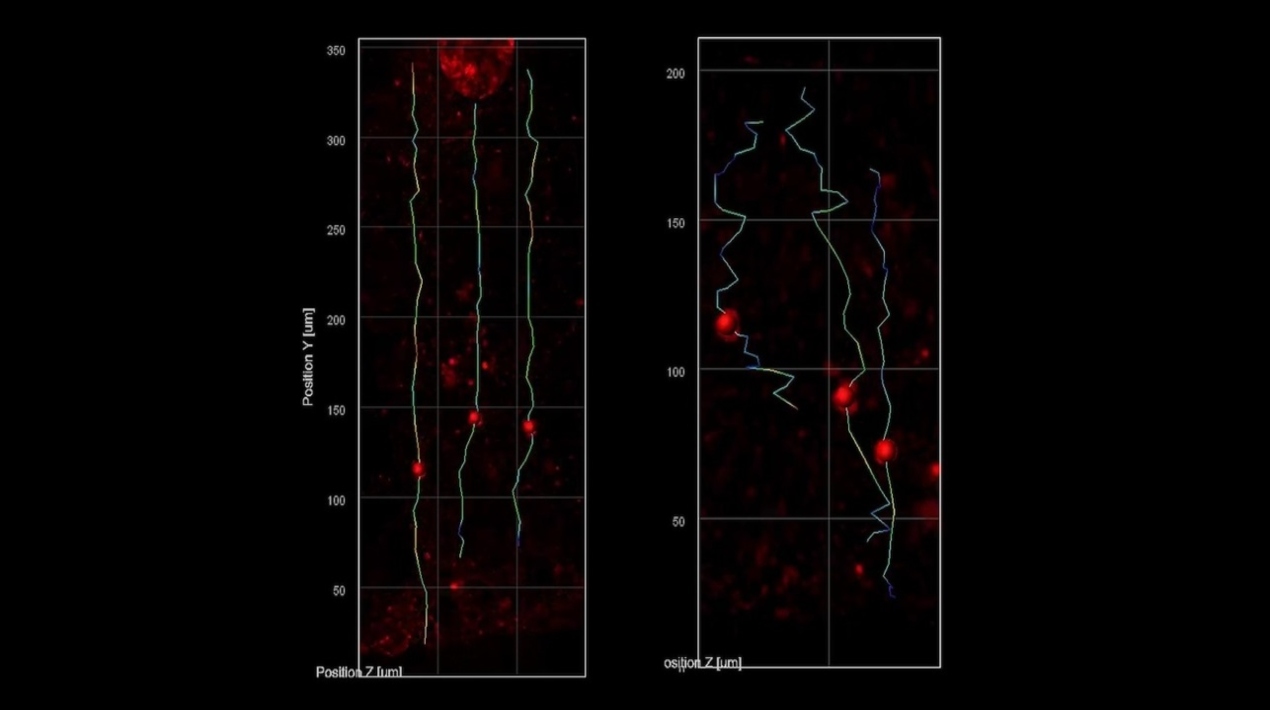
They were able to follow the migration of developing neurons as they moved from the inner edge, which is like the cortex of the brain, to the rim around ventricles in the mini-brains.
They observed that the nascent neurons in the mini-brains simulating Rett syndrome migrated more slowly and in meandering patterns than did the same cell types in mini-brains without the MECP2 mutation, which moved faster and in straighter lines.
The effects of these migration abnormalities are in line with what scientists believed to be happening in foetuses with Rett syndrome.
Researchers knew from post-mortem brains and brain imaging techniques that something goes awry during brain development in Rett syndrome, but it has been extraordinarily difficult to determine what and why; therefore, the technique has enabled them to directly visualise a critical component.
Due to its sensitivity to changes in the refractive index of materials, THG can scan tissues without labelling. Consequently, it dissolves the borders between biological structures such as blood vessels, cell membranes, and extracellular spaces.
The researchers were also able to distinguish clearly between the ventricular zone (the region around the ventricles where the new-born neurons arise) and the cortical plate because neural forms change as they develop (an area that mature neurons settle into). Additionally, it was quite simple to separate multiple ventricles into separate regions and resolve them.
















